
619 EDTA Antikoagulantien US Blood Tissue (Biology)
It is a popular chemical that goes by several names and is frequently utilized in medicinal and industrial applications. Ferdinand Munz was the first to synthesize this chemical in 1935. It is a colorless, crystalline, slightly soluble organic molecule utilized in biology and inorganic chemistry. It is a chelating agent.

What is EDTA? What is its denticity?
Scope and Significance Ethylenediaminetetraacetic acid (EDTA) is a well known metal-chelating agent, extensively used for the treatment of patients who have been poisoned with heavy metal ions such as mercury and lead.
PPT EDTA Titrations Chapter 13 PowerPoint Presentation, free download ID6757871
In this chapter, we introduce EDTA (Extensive de novo TE Annotator), a new comprehensive pipeline composed of high-quality tools to identify and annotate all types of TEs. The development of EDTA is based on the benchmarking results of a collection of TE annotation methods. The selected programs are evaluated by their ability to identify true.

The molecule structure of calcium chelated with eDTa and Pa.... Download Scientific Diagram
Applications- EDTA : EDTA is a popular chelating agent for divalent ions, that is widely used in biochemistry, molecular biology and cell biology. EDTA is an abbreviation for EthyleneDiamineTetraAcetic ac (and many other related molecules). EDTA is an amino acid widely used to sequester di- and trivalent metal ions (Ca2+ and Mg2+ for example).

EDTA Full Form Acid
TE buffer is a commonly used buffer solution in molecular biology, especially in procedures involving DNA, cDNA or RNA. "TE" is derived from its components: Tris, a common pH buffer, and EDTA, a molecule that chelates cations like Mg 2+. The purpose of TE buffer is to solubilize DNA or RNA, while protecting it from degradation. Recipe

Disodium Edetate Disodium EDTA Drug Molecule. Skeletal Formula. Stock Vector Illustration of
TAE buffer TAE buffer is a buffer solution containing a mixture of Tris base, acetic acid and EDTA. In molecular biology, it is used in agarose electrophoresis typically for the separation of nucleic acids such as DNA and RNA. [1] It is made up of Tris-acetate buffer, usually at pH 8.3, and EDTA, which sequesters divalent cations.

EDTA Full Form, Structure and Form, Uses, Formula
1. Introduction. Divalent metal ions including Mg 2+, Cu 2+, Fe 2+, Mn 2+, Ni 2+, and Zn 2+ play prominent roles in enzymatic catalysis. The small organic compound ethylene diamine tetraacetic acid or commonly known as EDTA is frequently used to chelate these metal ions to investigate enzyme function in the absence of metal co-factors.

Sigma Aldrich EDTA FOR MOLECULAR BIOLOGY Cas 6381926 Sigma Aldrich
Sequencing technology and assembly algorithms have matured to the point that high-quality de novo assembly is possible for large, repetitive genomes. Current assemblies traverse transposable elements (TEs) and provide an opportunity for comprehensive annotation of TEs. Numerous methods exist for annotation of each class of TEs, but their relative performances have not been systematically compared.

PPT EDTA Titrations PowerPoint Presentation ID234018
The ADI for humans as set by JEFCA is 1.9 mg EDTA/kg bw (or 2.5 mg Ca-EDTA/kg bw), corresponding to 23.37 mg EDTA/kg bw for mice and is slightly higher than the one we used (21 mg/kg bw) 31.

EDTA is a ligand.
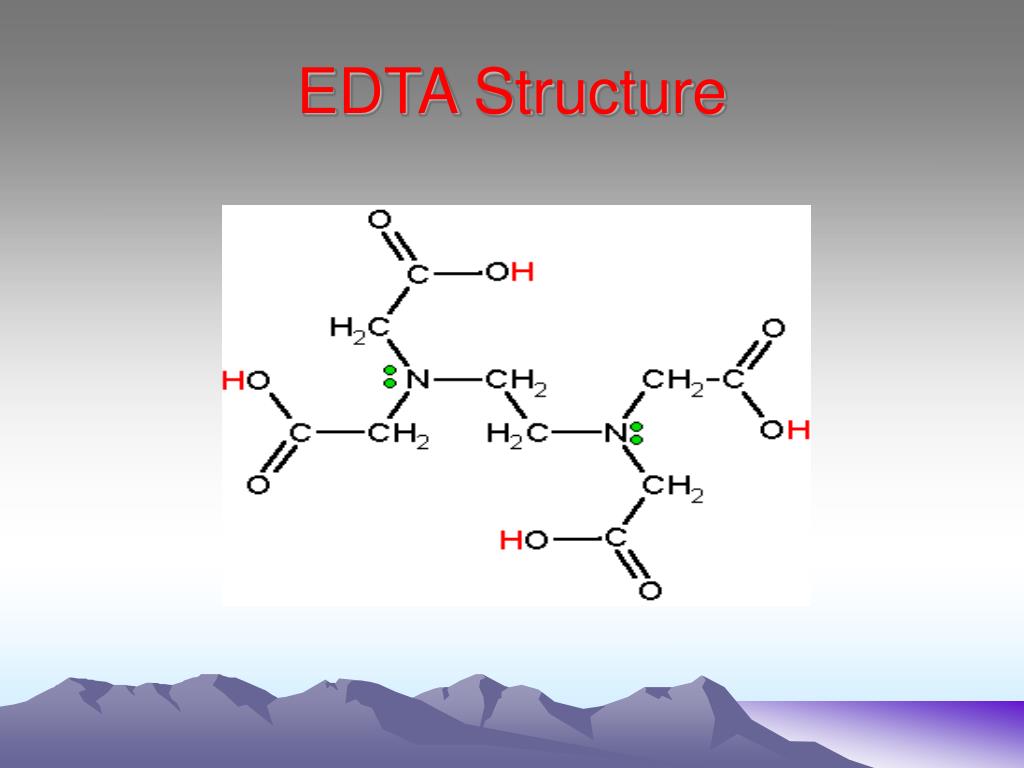
Ethylenediamine tetraacetic acid (EDTA) is a polyprotic acid containing four carboxylic acid groups and two amine groups with lone-pair electrons that chelate calcium and several other metal ions.
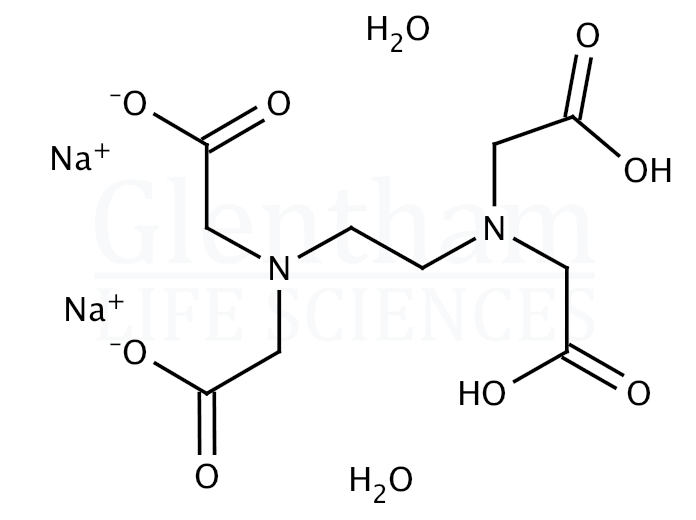
EDTA disodium salt dihydrate (CAS 6381926) Glentham Life Sciences
Popular answers (1) Michael J. Benedik Hamad bin Khalifa University EDTA is a commonly used chelating agent in molecular because it is a very effective chelator of Magnesium. Most nucleases.

Complexes formation. The figure 2 shows the EDTA ligand binding to a... Download Scientific
Biologically important ions such as Ca, K, Mg, Fe, and Zn play major roles in numerous biological processes, and their homeostatic balance is necessary for the maintenance of cellular activities. Sudden and severe loss in homeostasis of just one biologically important ion can cause a cascade of negative effects. The ability to quickly, accurately, and reliably quantify biologically important.
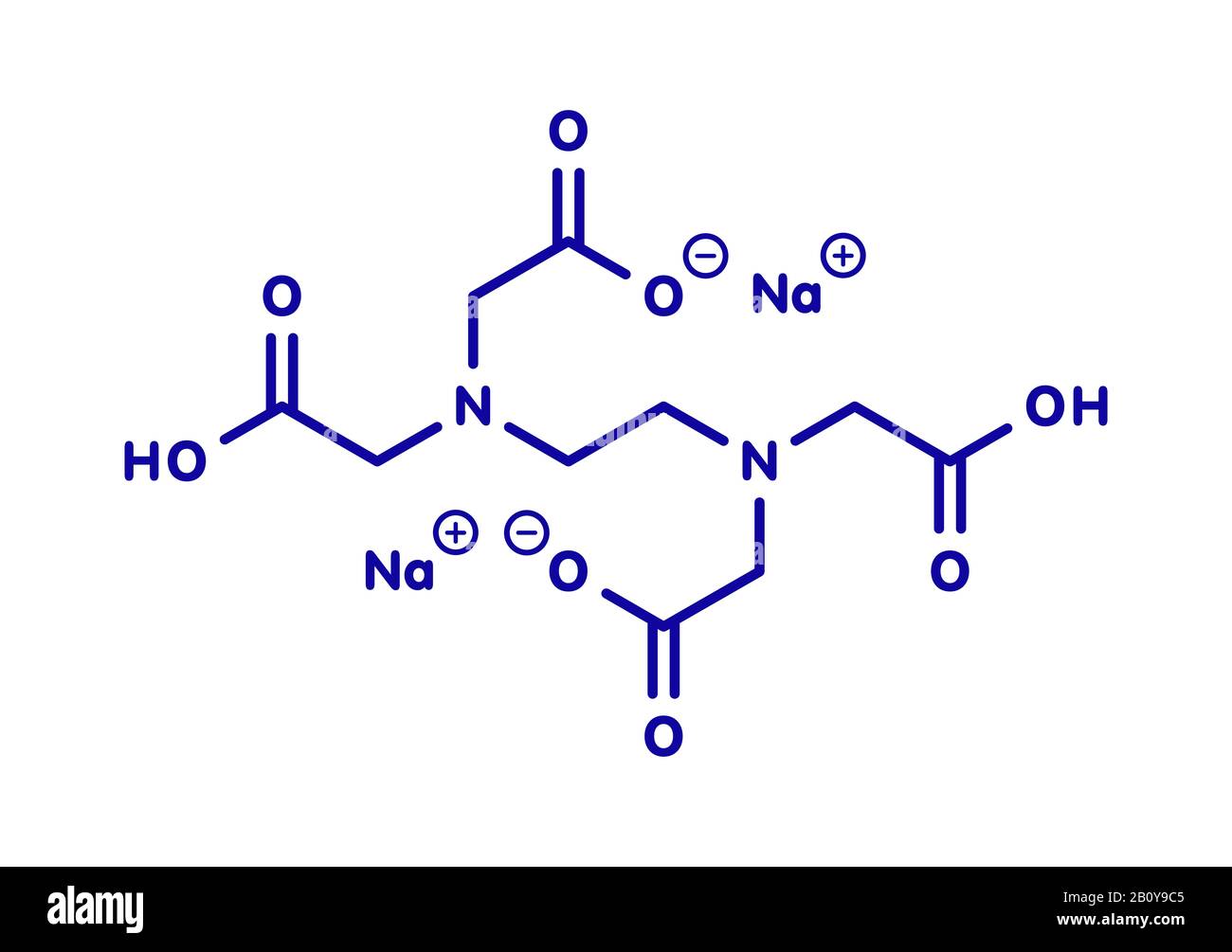
Disodium EDTA molecule, illustration Stock Photo Alamy
This increased attention to EDTA resulted in studies of EDTA used as an antioxidant occurring in more recent years. The review contained three studies where EDTA was used as an antioxidant in order to treat psoriasis [19-20,22]. Another study in the review utilized EDTA as a chemical treatment in order to cause a decrease of beta lipoproteins .

PPT EDTA Titrations Chapter 13 PowerPoint Presentation, free download ID6757871
Ethylenediaminetetraacetic acid (EDTA) is a medication used in the management and treatment of heavy metal toxicity. It is in the chelating class of drugs. This activity outlines and reviews the indications, actions, and contraindications for EDTA as a valuable agent in managing lead toxicity.

What is the Full Form of EDTA? I Leverage Edu
Chelation Therapy Jeanne A. Drisko MD, in Integrative Medicine (Fourth Edition), 2018 EDTA EDTA is poorly absorbed from the GI tract (<5%) and, as a consequence, should only be administered by a parenteral route. 4 It is primarily distributed in extracellular fluids, which limit its capacity to chelate intracellular metals.

EDTA Full Form Ethylene Diamine TetraAcetic Acid StudyWoo
Ethylenediaminetetraacetic acid ( EDTA ), also called edetic acid after its own abbreviation, is an aminopolycarboxylic acid with the formula [CH 2 N (CH 2 CO 2 H) 2] 2. This white, water-insoluble solid is widely used to bind to iron (Fe 2+ /Fe 3+) and calcium ions (Ca 2+ ), forming water-soluble complexes even at neutral pH.
